But as I explain, the study’s headline conclusion is highly misleading. The authors obscure the real story with mathematical sleight-of-hand that misrepresents the underlying data and overlooks the true nature of today’s pharmaceutical innovations.
Most notably, the authors discount the fact that the most expensive new drugs treat ultra-rare conditions affecting extraordinarily small patient populations. Their policy recommendations would therefore have a devastating impact on these patients and their hope for treatments and cures.
As Nobel prize winner Ronald Coase observed: “If you torture the data long enough, it will confess to anything.” Read on and consider what the data are really saying.
READ ME
As always, I encourage you to review the original source material for yourself. Here are the key materials:
- Prices for New Drugs Are Rising 20 Percent a Year. Congress Needs to Act., The New York Times
INDEX GAMES
The study purports to show that launch prices of new drugs have grown over time.
In general, this is true. As the authors accurately note: “The proportion of drugs priced at $150000 per year or more was 9% (18/197) in 2008-2013 and 47% (42/89) in 2020-2021.”
However, the authors make a crucial but subtle decision when computing an index to measure the change in prices over time. This decision renders their results misleading to the average reader and policy maker.
To study launch prices over time, the authors created an equally weighted price index of all new drugs that happened to be approved in a given year. Each product approved in a given year had the same contribution in that year’s average, regardless of utilization (population treated, number of prescriptions, etc.).
That’s simply not the correct way to calculate an accurate price index. This method wildly overstates average prices, by overweighting costly new drugs that treat ultra-rare conditions. It's like creating a consumer price index that simply averages the prices of a banana, a TV, and a Rolls Royce.
To understand the problem, consider a simple example with three orphan drugs launched in the same year with the following prices and patient populations:
- $1,000,000 product treating 1,000 patients
- $100,000 product treating 10,000 patients
- $10,000 product treating 100,000 patients
The methodology in the JAMA paper would provide the following summary statistics:
- Average price = $370,000
- Median price = $100,000
- Average price = $27,027
- Median price = $10,000
I don’t have access to the underlying data, so I can’t replicate the results or attempt to compute a proper index. But as far as I know, every news story about the JAMA study missed the mathematical sleight-of-hand behind the use of an unweighted index.
TORTURING THE DATA
The authors made another critical choice that distorted the measurement of growth rates.
Consider that the headline of their New York Times op-ed blared that "Prices for New Drugs Are Rising 20 Percent a Year." In the JAMA article, they described the rate of prices change from 2008 to 2021 as follows: “Unadjusted mean launch prices increased exponentially by 20.4% per year (95% CI, 15.3%-25.8% per year).”
However, both statements are a vast oversimplification of what the data actually show.
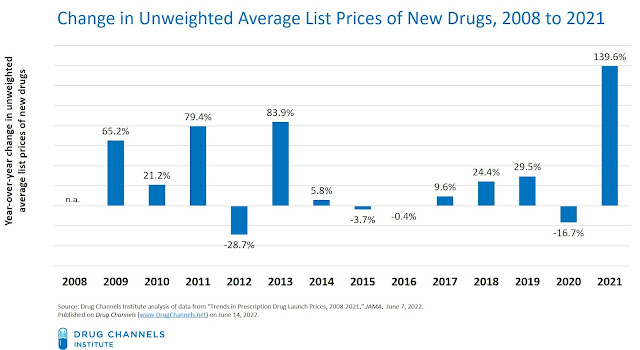
Dr. Rome, the study’s lead author, was kind enough to share the list price data behind the figure included in the JAMA article. Here are the year-over-year changes in average launch list prices, computed using the misleading unweighted methodology described above:
[Click to Enlarge]
As the chart above demonstrates, the “20% per year” story does not look quite so clear. Even a careful reader of the JAMA article would have been hard pressed to detect that year-over-year average prices (using the unweighted average) actually dropped in 4 of the 13 years studied!
The average growth rate was computed using the final year (2021) figure. However, the apparent spike reflected the approval of multiple ultra-rare therapies. This FiercePharma article notes such drugs as Eiger BioPharmaceuticals’ Zokinvy, which has a list price above $1 million. However, only 20 U.S. patients have been identified as candidates for this therapy.
The New York Times opinion piece further claims: “The rapid growth in prices is only partly attributable to the introduction of more complex medicines, such as injectables and biologics, or drugs focused on treating rare diseases.”
But that’s not an accurate description of their actual methodology. In the JAMA paper, the authors statistically account for the effect of patient population size with a variable that measures whether a drug treated a condition affecting fewer than 200,000 people. (See footnote f in the study’s table.) That’s fewer than 1 in 1,600 people within the U.S.
That doesn't fully account for the small populations being treated by cell and gene therapies and other drugs treating ultra-rare conditions. These drugs typically target patient populations with fewer than 5,000 people, i.e., less than 1 in 66,000 people.
RARE ILLOGIC
The authors draw the following conclusions from their analyses:
“Negotiating drug prices will not harm innovation. In fact, the status quo of allowing drug companies to freely set prices incentivizes development of many products that do not offer important therapeutic advances. These less innovative drugs offer low financial risk to manufacturers because they are based on existing products or work via similar biochemical pathways.”These statements are not supported by their data. Expensive drugs for ultra-rare conditions have a minimal impact on overall drug spending, despite their headline-grabbing price tags. Consider such expensive but highly effective CAR-T treatments as Novartis’ Kymriah and Gilead’s Yescarta. Both were approved over the past few years, but have so far generated combined net U.S. sales of only $636 million in 2021. Even ICER analyses conceded that these drugs' high prices "appear to be priced in alignment with their clinical value."
In reality, no one really knows what will happen if selling prices for ultra-rare conditions were sharply reduced. Note that the Congressional Budget Office’s (CBO) models of government price negotiation and its impact on drug development can't tell us much, because the models are specified using older research studies from the time before the launch of ultra-rare therapies.
With their JAMA study and accompanying op-ed, Dr. Rome, Mr. Egilman, and Dr. Kesselheim have therefore launched a new attack on patients with ultra-rare conditions. Let’s all hope that cooler heads will evaluate the evidence and not destroy the innovation ecosystem developing rare disease treatments.
CORRECTION: An earlier version of this article misstated the mean and median values for the weighted average prices.


No comments:
Post a Comment